when the electron's principal quantum number n = ∞. In the formulas for energy of electrons at various levels given below in an atom, the zero point for energy is set when the electron in question has completely left the atom, i.e. The modern quantum mechanical theory giving an explanation of these energy levels in terms of the Schrödinger equation was advanced by Erwin Schrödinger and Werner Heisenberg in 1926.Ītoms Intrinsic energy levels The notion of energy levels was proposed in 1913 by Danish physicist Niels Bohr in the Bohr theory of the atom. The first evidence of quantization in atoms was the observation of spectral lines in light from the sun in the early 1800s by Joseph von Fraunhofer and William Hyde Wollaston. Measurement of the possible energy levels of an object is called spectroscopy. A measurement of the energy results in the collapse of the wavefunction, which results in a new state that consists of just a single energy state. Elementary examples that show mathematically how energy levels come about are the particle in a box and the quantum harmonic oscillator.Īny superposition ( linear combination) of energy states is also a quantum state, but such states change with time and do not have well-defined energies. Informally, these states correspond to a whole number of wavelengths of the wavefunction along a closed path (a path that ends where it started), such as a circular orbit around an atom, where the number of wavelengths gives the type of atomic orbital (0 for s-orbitals, 1 for p-orbitals and so on). States having well-defined energies are called stationary states because they are the states that do not change in time. For a confined particle such as an electron in an atom, the wave functions that have well defined energies have the form of a standing wave. Quantized energy levels result from the wave behavior of particles, which gives a relationship between a particle's energy and its wavelength. 
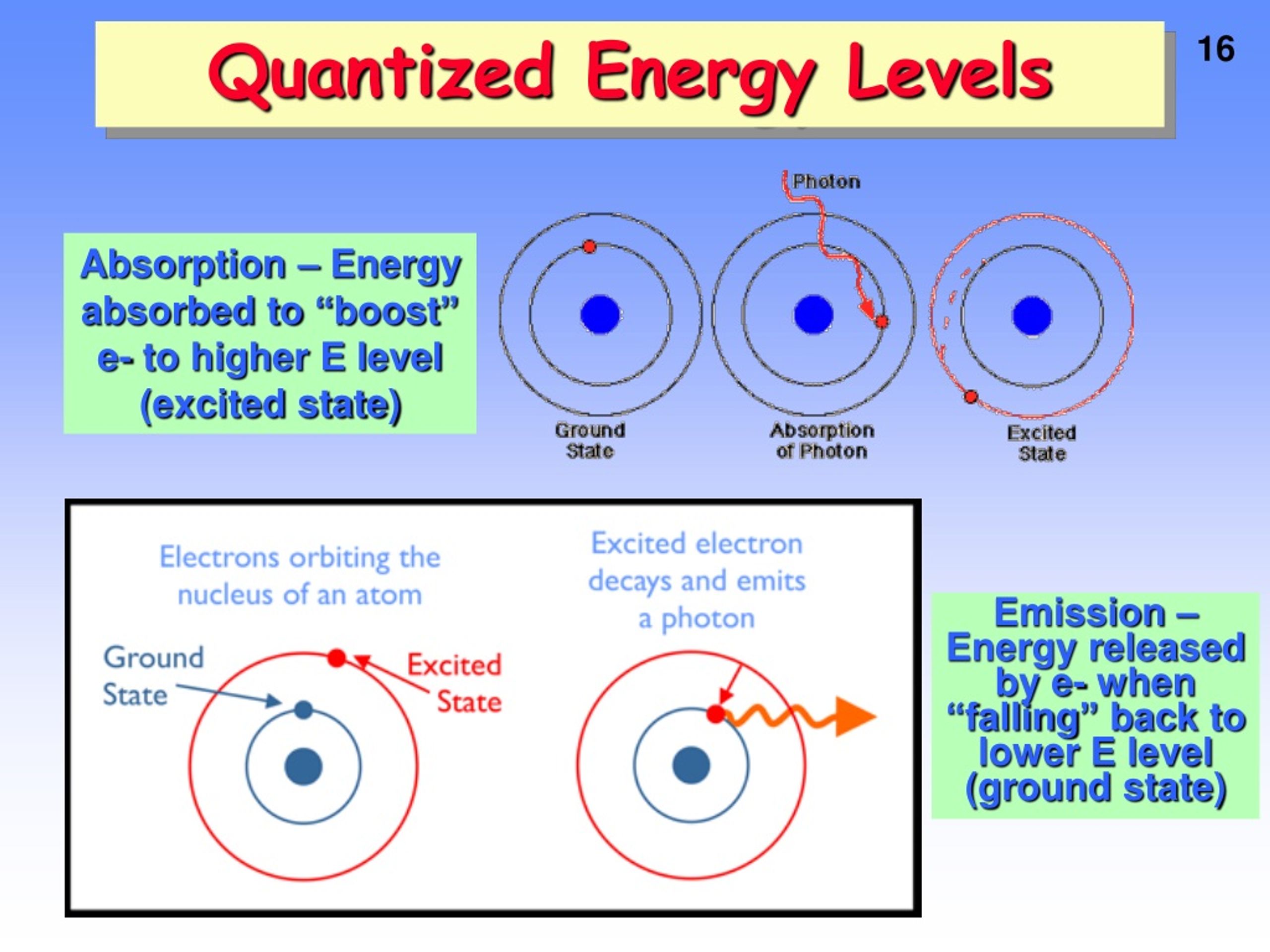
Each stationary state defines a specific energy level of the atom. Wavefunctions of a hydrogen atom, showing the probability of finding the electron in the space around the nucleus. An energy level is regarded as degenerate if there is more than one measurable quantum mechanical state associated with it. If it is at a higher energy level, it is said to be excited, or any electrons that have higher energy than the ground state are excited. If an atom, ion, or molecule is at the lowest possible energy level, it and its electrons are said to be in the ground state. If the potential energy is set to zero at infinite distance from the atomic nucleus or molecule, the usual convention, then bound electron states have negative potential energy. (See Madelung rule for more details.) For an explanation of why electrons exist in these shells see electron configuration. 
However, this is not a strict requirement: atoms may have two or even three incomplete outer shells. Since electrons are electrically attracted to the nucleus, an atom's electrons will generally occupy outer shells only if the more inner shells have already been completely filled by other electrons. The general formula is that the nth shell can in principle hold up to 2 n 2 electrons. The shells correspond with the principal quantum numbers ( n = 1, 2, 3, 4 .) or are labeled alphabetically with letters used in the X-ray notation (K, L, M, N.).Įach shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The closest shell to the nucleus is called the " 1 shell" (also called "K shell"), followed by the " 2 shell" (or "L shell"), then the " 3 shell" (or "M shell"), and so on farther and farther from the nucleus. In chemistry and atomic physics, an electron shell, or principal energy level, may be thought of as the orbit of one or more electrons around an atom's nucleus. The energy spectrum of a system with such discrete energy levels is said to be quantized. The term is commonly used for the energy levels of the electrons in atoms, ions, or molecules, which are bound by the electric field of the nucleus, but can also refer to energy levels of nuclei or vibrational or rotational energy levels in molecules. This contrasts with classical particles, which can have any amount of energy. A quantum mechanical system or particle that is bound-that is, confined spatially-can only take on certain discrete values of energy, called energy levels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
